pMVL Technology
Proprietary multivesicular liposome (pMVL) technology encapsulates drugs, preserving their molecular structure releasing them over a desired period of time.
How pMVL works
The pMVL carrier matrix consists of microscopic, spherical, lipid-based particles composed of a honeycomb of numerous, non-concentric, internal aqueous chambers containing the encapsulated active agent. Each chamber is separated from adjacent chambers by lipid membranes. Following injection, the pMVL particles release the active agent over an extended period of time due to erosion and/or reorganization of the lipid membranes.
Features and benefits
- pMVL offers flexible delivery and can be designed to offer an immediate-release dose followed by sustained delivery
- The ready-to-use aqueous solution works with narrow gauge needles and pen systems
- pMVL membrane components are natural, well-tolerated, and cleared by normal metabolic pathways
- pMVL permits systemic delivery of medications by releasing the drug into the bloodstream via the interstitial space
- Locally, pMVL can release the active agent into a body compartment, such as a wound
An advanced drug delivery platform
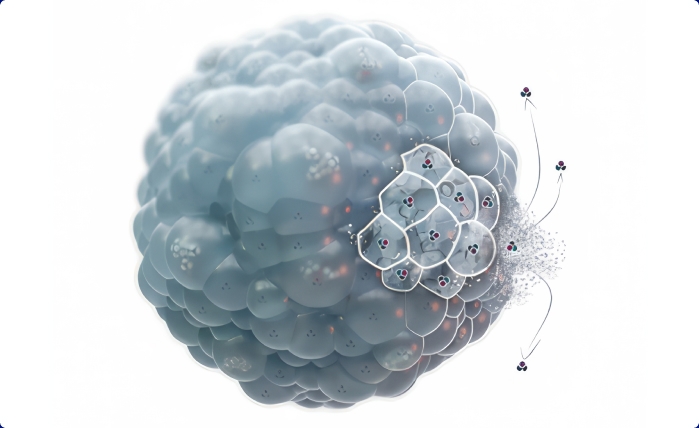
to deliver controlled levels of active agents.1
of naturally occurring, biocompatible lipids2-4
the active agent in a suspension of multivesicular liposomes
the active agent over time1
Discover the potential for pMVL
pMVL applications
Our pMVL extended-release drug delivery technology can be applied to both small and large molecules, potentially improving patient care by providing a unique solution for medications that:
- Need to be administered by continuous infusion or frequent multiple injections
- Have a short duration of action or problematic side effects associated with peak concentrations
We have demonstrated that small and large molecule compounds can successfully be encapsulated into pMVL, including:
- Traditional injectable pharmaceuticals
- Proteins
- Peptides
- Antisense oligonucleotides
- DNA
Utilizing pMVL to extend pain relief
We are leveraging the proven safety, flexibility, and customizability of our pMVL technology for acute, sub-acute, and chronic pain applications. Our first application was encapsulating bupivacaine to control pain and reduce or eliminate the use of opioids post-surgery.
In addition, we are exploring the use of our pMVL technology in combination with other therapies to drive longer-term pain relief.
References
- Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536.
- Angst MS, Drover DR. Pharmacology of drugs formulated with DepoFoam: a sustained release drug delivery system for parenteral administration using multivesicular liposome technology. Clin Pharmacokinet. 2006;45(12):1153-1176.
- Kohn FR, Malkmus SA, Brownson EA, Rossi SS, Yaksh TL. Fate of the predominant phospholipid component of DepoFoam drug delivery matrix after intrathecal administration of sustained-release encapsulated cytarabine in rats. Drug Deliv. 1998;5(2):143-151.
- Richard BM, Newton P, Ott LR, et al. The safety of EXPAREL® (bupivacaine liposome injectable suspension) administered by peripheral nerve block in rabbits and dogs. J Drug Deliv. 2012;2012:962101.